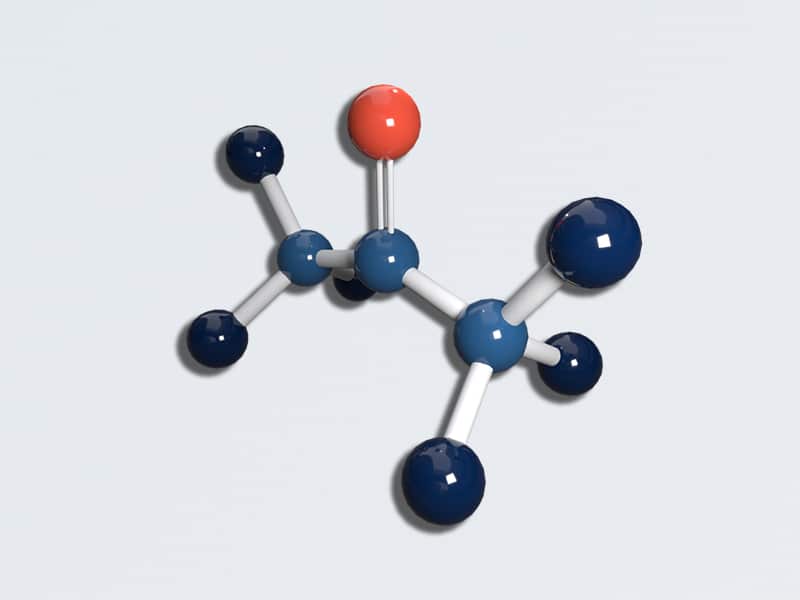
Acetone
Formula: C3H6O | CAS: 67-64-1
Acetone detection: Acetone poisoning occurs when there’s more acetone in your body than your liver can break down.
Acetone detection, Why do we use acetone?
The majority of the world’s acetone is used as a solvent, but it also has laboratory, medical, cosmetic and other uses. Acetone’s most well-known use is in nail salons, but it appears in many other industries:
- Printing – as a cleaning solvent
- Adhesives Manufacturing – most commonly in carpet adhesives
- Wood Stains and Varnishes – for varnish solvents – very common in households
- Paint Stripping – as a solvent, also common in households
- Polystyrene Manufacture – for polystyrene production
- Machinery Manufacture and Repair – as a cleaning solvent
- SBR Latex Production – as a solvent
- Electroplating – as a vapour degreaser and cold-cleaning solvent
When acetone is breathed in or ingested it can be absorbed into the body. This can cause headache, movement problems, tiredness slurred speech, nausea, vomiting and fitting. There may be a fruity smell on the breath. In severe cases there may be a swelling of the brain, unconsciousness, low body temperature and slowed breathing.
– GOV.UK
Affects of Short & Long term exposure to acetone
SHORT TERM EXPOSURE: (ACUTE)
Mild nervous system effects such as eye and respiratory irritation, mood swings, and nausea that abated soon after exposure ended were seen in humans breathing high concentrations of acetone. Accidental poisonings report similar nervous system effects of sluggishness and drowsiness that were not long lasting.
Humans exposed to high levels of acetone by inhalation experienced eye and nasal irritation. Exposure to somewhat lower concentrations did not cause any adverse health effects. In another study, groups of students were exposed by inhalation to acetone for six hours. At the higher concentrations, eye, nose and throat irritation were observed.
LONG TERM EXPOSURE: (CHRONIC)
Workers exposed by inhalation to acetone for three hours per day for seven to 15 years complained of respiratory tract irritation, dizziness, and loss of strength.
In animals exposed to very high doses of acetone in drinking water, effects on the blood indicating an anemic condition were reported.
Gas Factsheet
Everything you need to know about acetone detection
Our Gas Factsheets which is available to download below provides you with key information on the exposure limits and the locations of where potentially harmful gases can occur. We also share information on gas detection monitoring techniques and equipment that can help you manage gas detection in the workplace, for worker and site safety.

Acetone detection
Formula: C3H6O | CAS: 67-64-1
Synonyms: propan-2-one, d-acetone, dimethylcetone, isopropanal, methylketone, Sasetone, methyl-ketone, b-ketopropane, 2-propanal, methyl methyl ketone, dimethyl ketone, methyl ketone, dimethylformaldehyde, pyroacetic ether, beta-ketopropane, ketone propane, pyroacetic acid, dimethylketone, beta-ketopropane, UN1090, RCRA waste no. U002, d-acetone, dimethylcetone, isopropanal, methylketone, Sasetone, methyl-ketone, b-ketopropane, 2-propanal, methyl methyl ketone, 2-propanone, propanone
Acetone finds major use in the manufacture of methacrylate. Acetone is a widely used solvent, particularly for cellulose acetate and various resins, paints varnishes and lacquers.
Related Products
Download your Gas Factsheet
Simply complete the form below to obtain your Gas Factsheet on “Acetone”.








 United Kingdom
United Kingdom





