
The use of PID for Benzene detection
Rather than having to rely on human senses in a workplace setting it is advisable to use an appropriate form of quantitative monitoring; indeed the onus is on the employer to do the risk assessment. Remembering the old adage attributed to Lord Kelvin that you can’t manage what you don’t measure, there are methods published by the Health and Safety Executive in the UK and NIOSH in the USA that can be used to capture air samples for later analysis but by definition this occurs after exposure could have taken place. Therefore real-time methods are preferable which can range from fixed, permanent systems for fence line applications, hand-held devices for area measurements or confined space entry and most recently developed, personal monitors that can alert a worker of an immediate hazard.
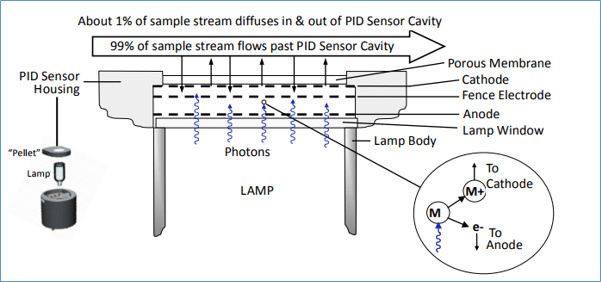
Collectively these devices are known as photoionisation detectors (PID) which gives a clue to their theory of operation. Figure 1 (below) is a schematic of an Ion Science Ltd PID sensor system. A UV lamp generates high-energy photons, which pass through the lamp window and a mesh electrode into the sensor chamber. Sample gas is pumped over the sensor and about 1% of it diffuses through a porous membrane filter into the other side of the sensor chamber. The inset on the lower right shows what happens on a molecular level. When a photon with enough energy strikes a molecule M, an electron (e-) is ejected. The M+ ion travels to the cathode and the electron travels to the anode, resulting in a current proportional to the gas concentration. The electrical current is amplified and displayed as a ppm or ppb concentration. Not all molecules can be ionised, thus, the major components of clean air, i.e., nitrogen, oxygen, carbon dioxide, argon, etc., do not cause a response, but most VOCs do give a response.
Three PID lamps are available with maximum photon energies, measured in electron-volts (eV), of 10.0 eV, 10.6 eV, and 11.7 eV.
Figure 2 (below) The illustration below shows the relationship between lamp energies expressed in electron Volts (eV) and a selection of VOC gases with Ionisation Energy (IE). The IE is the energy level required to Ionise each gas and create an electronic signal. Typically, PID sensors fitted with higher energy lamps can detect a wider range of gases. PID for Benzene detection – when detecting benzene however lower output lamps maybe chosen as other less harmful VOC’s are undetected.
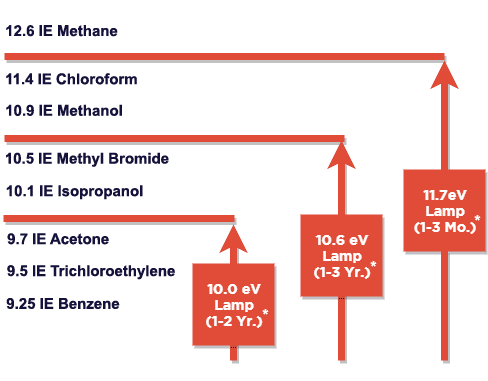
*Expected lamp life subject to use and application
The choice of lamp normally depends on the application.
From the lamps shown above 11.7 eV lamps detect the widest range of gases as it has the highest energy output. These lamps however can be difficult to manufacture so are generally more costly. The window of the 11.7 eV lamp is also brittle and lamp life can be reduced if used in high humidity environments. Although 11.7 eV lamps detect the widest range of gases the above limitations means they are only ever used when necessary.
The most readily available lamp has an ionisation energy of 10.6 eV and these are relatively straightforward to manufacture. They tend to be at a lower cost point and offer a longer life but this can be affected by the application.
10.0 eV lamps detect less gases than the others however in some circumstances this can be beneficial. A petrochemical environment may have many detectable VOC gases present, if trying to identify the level of benzene many other less harmful VOC’s are ignored as they are undetected, this is used as a type of screening.
Another term used to detect gases with similar IE levels as benzene is TACs (Total aromatic Compounds).
The partial screening effect of the 10.0 eV lamp combined with the filtering effect of a benzene pre-filter can result in the measurement of benzene specifically even if VOC and/or TAC gases are present.
Download our Guide: The Basics of Benzene








 United Kingdom
United Kingdom





